Last Week's Guest Speaker:
Rod Slater,
"Hydrogen Powered Vehicles"
Guest Speaker Rod Slater
Picture Doug Worthington
Picture Doug Worthington
I first became aware of Hydrogen fuel in 2004 when as president of the RAC WA I attended a three day conference. At that conference I happened to be sitting next to an American couple and his task at NASA was to calculate the liquid hydrogen fuel load to power the rocket to the moon and back in 1969.
Now transportation from hydrogen derived from h2o, water, is the way forward as hydrogen is the most abundant element in the universe. We are moving from fossil fuel to 2030 target for electric vehicles.
The problem for electric vehicles is that of battery life, the cost of replacement of the batteries, the time taken to recharge a fully electric vehicle, and the limited distance of travel for the vehicle.
In 1900 electric vehicles dominated because the fossil fuel cars were noisy with no mufflers, and hard to start until the starter motor. In 1920 when fossil fuel cars had addressed these issues there were no electric cars.
The current challenge with electric vehicles is that electricity is derived from fossil fuels and the challenge is to have the electricity from renewable energy.
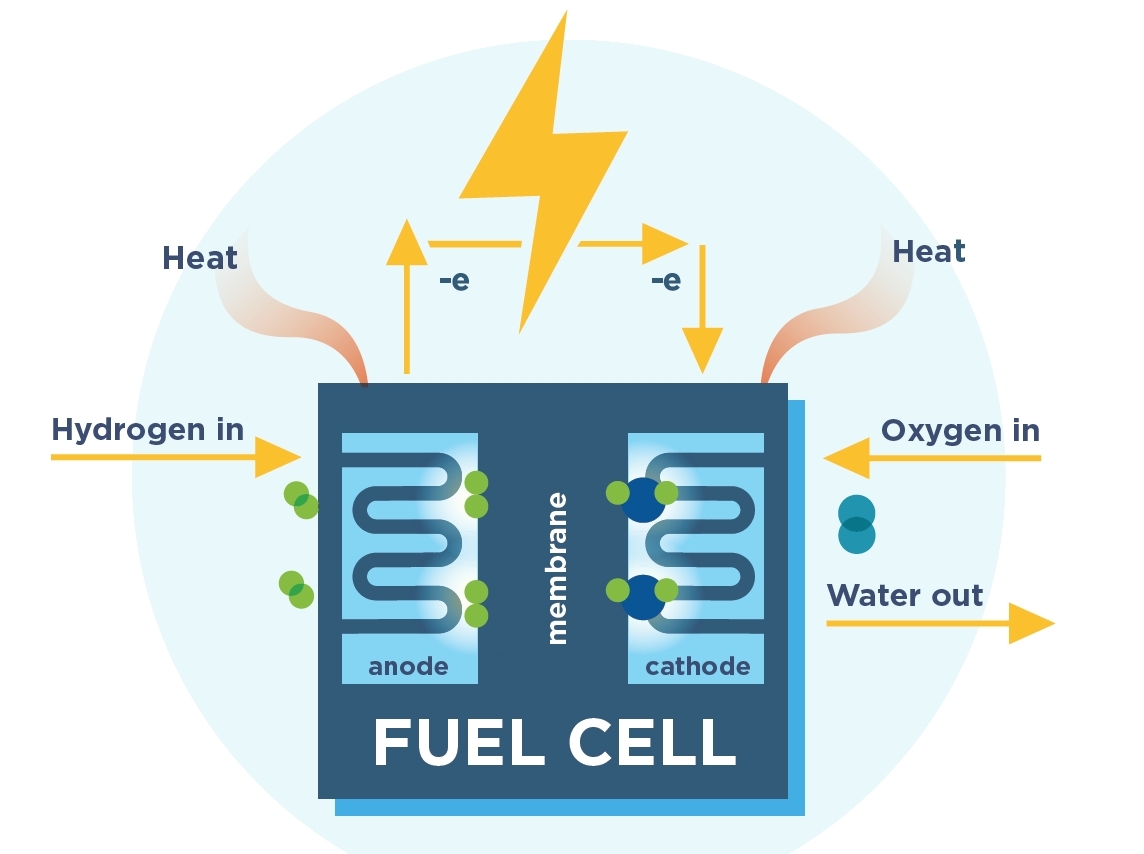
The hydrogen fuel cell is solid with no moving parts. The hydrogen is taken from a storage tank in the vehicle and mixes with air to produce electricity and water vapour.
[Source: http://www.fchea.org/fuelcells]
A fuel cell is composed of an anode, a cathode, and an electrolyte membrane. A fuel cell works by passing hydrogen through the anode of a fuel cell and oxygen through the cathode. At the anode site, the hydrogen molecules are split into electrons and protons. The protons pass through the electrolyte membrane, while the electrons are forced through a circuit, generating an electric current and excess heat. At the cathode, the protons, electrons, and oxygen combine to produce water molecules.
Due to their high efficiency, fuel cells are very clean, with their only by-products being electricity, excess heat, and water. In addition, as fuel cells do not have any moving parts, they operate near-silently.
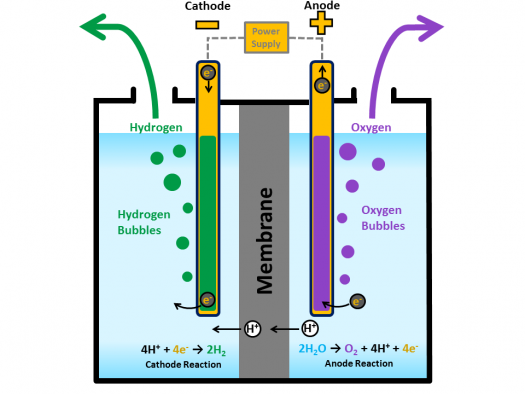
Extract hydrogen from water via an electric current. Electrolysis is the technical name for using electricity to split water into its constituent elements, hydrogen and oxygen. The splitting of water is accomplished by passing an electric current through water. The electricity enters the water at the cathode, a negatively charged terminal, passes through the water and exists via the anode, the positively charged terminal. The hydrogen is collected at the cathode and the oxygen is collected at the anode. Electrolysis produces very pure hydrogen from water.
Electrolysis is very expensive. The electrical inputs required to split the water into hydrogen and oxygen account for about 80% of the cost of hydrogen generation. Potentially, electrolysis, when coupled with a renewable energy source, can provide a completely clean and renewable source of energy. So there would need to be a windfarm next to the ocean to produce the electricity to remove the hydrogen from the water.
[Source: https://www.energy.gov/eere/fuelcells/hydrogen-production-electrolysis]
Economically hydrogen is similar to petroleum. There is the plan for a hydrogen highway from Port Headland to Bunbury through the State Government with three companies.
A vehicle can add more fuel cells for more power for a larger vehicle with each fuel cell producing 240 kilowatts per cell.
Chageover Dinner Photos
By Doug Worthington

Glenda as master of ceremonies

Incoming President Allan

Girrawheen Senior High School represenattives past and current
Congratulations to Greg Hunter
Outgoing President Greg was awarded Rotary’s highest honour, Paul Harris Fellow, Greg is holding the medal.

Congratulations to Russel Miln
Russel from Girrawheen Senior High School was awarded Rotary’s highest honour, Paul Harris Fellow, Russle with District Governor Graham.

.jpg)








